The Cutting Edge of Medical Technology Content, Community & Collaboration
Dissolution testing is a very important tool that determines and help understand the performance and effectiveness of oral solid dosage forms. It is significant for the field of medicine because if a drug has to be effective, it must be released first from the product form, and it should then be allowed to get dissolved in the gastrointestinal fluids. This is the first step that leads to the next important phase, that of the dosage’s absorption into the bloodstream. This points to the fact that dissolution from the dosage form is a major determinant of the rate and extent to which the drug gets absorbed by the body.
Drug dissolution testing is very important during the development of drugs and drug formulations. It helps to determine if the right concentration of the drug reaches the desired or expected locus of action. This makes the investigation of the factors which affect drug absorption into the human blood flow when a drug product is taken orally important.
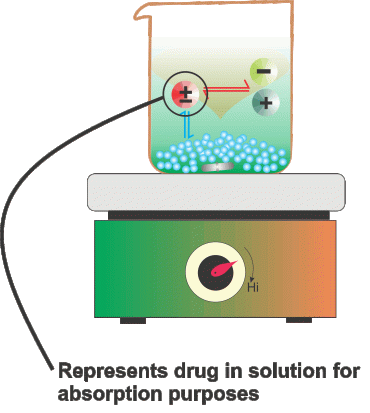
The usual method of measurement of drug absorption is in vivo, or, the body of a living being such as a human or animal. Time blood plasma concentration profiles of drugs after oral administration constitute an important in vivo parameter. In-vitro investigations are carried out for identifying the parameters involved in drug absorption. These are investigations that are conducted in a controlled and simulated environment that resembles biological conditions closely.
Thorough learning of drug dissolution
An important seminar from GlobalCompliancePanel, a leading provider of professional trainings for all the areas of regulatory compliance, will offer valuable learning on all the aspects of drug dissolution testing and explain the ways of establishing plasma drug levels in humans.
At this two-day seminar, Dr. Saeed Qureshi, who has worked as a research scientist with Health Canada and is an internationally known expert on the subject whose expertise spans the areas of drug dissolution testing, pharmacokinetics, biopharmaceutics and analytical chemistry as related to animal and human studies for developing and evaluating pharmaceutical products; will be the Director.
In order to gain the benefit of learning from this world-renowned expert, please enroll for this seminar by visiting Drug dissolution testing and establishing plasma drug levels in humans. This course has been pre-approved by RAPS as eligible for up to 12 credits towards a participant’s RAC recertification upon full completion.
All aspects of drug dissolution and establishing plasma drug levels

This seminar will provide its participants a unique opportunity to learn scientifically valid drug dissolution testing and establishing plasma drug levels. Lab personnel take several approaches to conduct dissolution testing using different apparatuses and methods. This makes section of an appropriate apparatus and method confusing and challenging. Dr. Qureshi will offer relevant pharmacokinetics and physiological background that is aimed at making this choice easier and intuitive. He will use simple and clear language in helping participants understand how to select or develop a dissolution method. He will describe the theoretical aspect of the drug dissolution testing, including method development, in detail. He will explain the pros and cons of different approaches.
Another important area that Dr. Qureshi will address is in vitro-in vivo correlation (IVIVC). He will address the particular issue of the use of the concepts of convolution/deconvolution and IVIVC in providing an estimate/prediction of expected drug levels in humans through drug dissolution testing. This approach has met with limited success. Dr. Qureshi will explain the reasons for this and suggest alternative approaches and will offer an explanation of the underlying scientific principles involved in convolution, deconvolution and IVIVC techniques with simple practical examples. He will describe a unique and simple approach based on convolution technique using spreadsheet software.
He will show in vitro drug dissolution testing and convolution/deconvolution techniques for predicting plasma drug levels using the principles of pharmacokinetics and physiology. Dr. Qureshi will cover the following main areas at this seminar, with its relevant subtopics:
- Physiological and Pharmacokinetic Principles
- Drug Dissolution Testing
- Linking Dissolution Results to Plasma Drug Levels.
Personnel who work in various levels of the areas of Pharmaceutical Development, setting up analytical methods (pharmacopeial, regulatory or in-house developed), R & D (both analytical and formulation), Project Management, Quality Control, Quality Assurance, and Regulatory Affairs will benefit enormously from this learning.
Views: 135
Comment
© 2025 Created by CC-Conrad Clyburn-MedForeSight.
Powered by
![]()
You need to be a member of MedTech I.Q. to add comments!
Join MedTech I.Q.