Want to enhance your regulatory compliance career by learning a new course? All that it costs is $10. Yes, GlobalCompliancePanel, a provider of professional trainings, is offering hundreds of high value regulatory compliance courses for a mere $10…
The Cutting Edge of Medical Technology Content, Community & Collaboration
All Blog Posts Tagged 'Medical' (125)
 Learn any professional courses for $10 only
Learn any professional courses for $10 only
Added by John Robinson on August 18, 2017 at 7:08am — No Comments
 How Americans get their health insurance
How Americans get their health insurance
With Obamacare firmly in the crosshairs of Republican lawmakers, the debate around U.S. healthcare is at a fever pitch.
While there is no shortage of opinions on the best route forward, the timeliness of the debate also gives us an interesting chance to dive into some of the numbers around healthcare – namely how people even get coverage in the first place.
How Americans get healthcare
The following infographic shows a breakdown of how…
Added by John Robinson on August 9, 2017 at 5:59am — No Comments
 How to create processes and procedures to implement them?
How to create processes and procedures to implement them?
Product Risk Management is a critical aspect of ensuring medical devices are safe and effective for intended uses. This course will help you understand the regulatory requirements, including ISO14971.
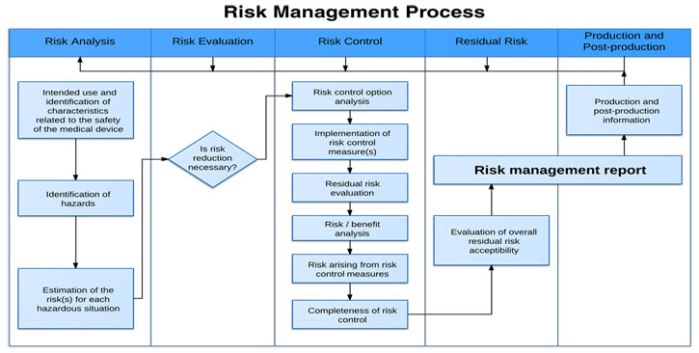
You’ll learn techniques that can help you identify hazards and potential harms. You’ll learn how to mitigate risk and effectively monitor risk to ensure your customers receive safe and effective products. A rigorous…
ContinueAdded by Adam Fleming on July 19, 2017 at 5:47am — No Comments
 Steps to IEC 60601-1 approval
Steps to IEC 60601-1 approval
The 60601-1 is a standard that relates to the safety of medical electrical equipment. This harmonized standard is recognized by most countries around the world. Existing and new medical devices have to comply with the requirements set out in IEC 60601-1.
The IEC 60601-1 is going through its latest revision, its 3rd edition,…
ContinueAdded by Adam Fleming on July 14, 2017 at 6:27am — No Comments
 How to control those risks and monitor the effectiveness of the controls put in place
How to control those risks and monitor the effectiveness of the controls put in place
Overview of this session Product Risk Management is a critical aspect of ensuring medical devices are safe and effective for intended uses. This course will help you understand the regulatory requirements, including ISO14971, and how to create processes and procedures to implement them.
You’ll learn techniques that can help you identify hazards and potential harms. You’ll learn how to mitigate risk and effectively monitor risk to ensure your customers receive safe and effective…
ContinueAdded by Adam Fleming on July 13, 2017 at 5:43am — No Comments
 Advantages and disadvantages of Electronic Health Records
Advantages and disadvantages of Electronic Health Records
Electronic Health Records (EHR) or electronic medical records (EMR), as they are called, are of enormous use in the fields of healthcare and medical sciences. They have a number of features that enable the patient; the medical professional and the healthcare provider have complete and unimaginably easy access to all important records that relate to the patient.
A direct result of the…
ContinueAdded by Roger Steven on June 29, 2017 at 7:52am — No Comments
 Maximizing profits and patient outcomes in the backdrop of declining reimbursements
Maximizing profits and patient outcomes in the backdrop of declining reimbursements
In the situation of falling revenues from Medicare billing, many practitioners are worried about the state of their future earnings. A very tangible factor that will help them tide over this problem is often overlooked: the Durable Medical Equipment (DME) program.
The DME program is a very reliable and long term source of income for Medicare practitioners. Yet, this is not considered seriously by most of them because of the various misconceptions that are associated…
Added by Adam Fleming on June 13, 2017 at 5:46am — No Comments
 Why IEC 60825 certification cannot be substituted for 21CFR1040
Why IEC 60825 certification cannot be substituted for 21CFR1040
With a few exceptions, it is necessary for manufacturers, system integrators and importers of lasers or laser containing products to implement best practices for compliance with FDA 21 CFR 1040. This is because The Center for Devices and Radiological Health (CDRH) requests documentation using the current version of their Form 3632 Guide for Preparing Product Reports for Lasers or Products Containing Lasers for laser self-certification submittals, which have to comply with…
Added by Adam Fleming on May 24, 2017 at 6:31am — No Comments
 Seminar Calendar of Upcoming Courses - June to July - 2017
Seminar Calendar of Upcoming Courses - June to July - 2017
GlobalCompliancePanel’s seminars are a wonderful opportunity for professionals in the regulatory compliance areas to understand the latest happenings and updates in the…
ContinueAdded by John Robinson on May 24, 2017 at 5:51am — No Comments
 Webinar Calendar of Upcoming Courses - May to Jun 2017
Webinar Calendar of Upcoming Courses - May to Jun 2017

Below is the event description content:
Compliance4All webinars are just what professionals in the regulatory
compliance areas need for scaling up in their careers. With a collection of
the most erudite experts on regulatory compliance being available at a click
in the comfort of your preferred location; regulatory compliance could not
get any simpler and effective!
Compliance4All’s experts help you unravel all the knowledge you need…
Added by Adam Fleming on May 22, 2017 at 7:53am — No Comments
 Meeting labeling requirements of various drug products
Meeting labeling requirements of various drug products
Both the FDA and the European Medicines Agency (EMA) have regulations that cover the labeling requirements of both prescription and over-the-counter (OTC) drugs, cosmetics, generics, medical devices, nutraceuticals and other related products. These regulations have to be strictly complied with. 21 CFR under its various parts, the Federal Food, Drug, and…
Added by Adam Fleming on May 19, 2017 at 6:17am — No Comments
 Regulations governing how combination products are regulated
Regulations governing how combination products are regulated
The knowledge of combination products and their regulations is essential for regulatory professionals. This is because of the high proportion of combination products in the market, as well as the array of regulations that govern them.
21 CFR 3.2 (e) has a complete definition of a combination product. A combination product is one…
Added by Adam Fleming on May 17, 2017 at 6:40am — No Comments
 Transiting from paper records to electronic for pharmaceutical companies while being compliant
Transiting from paper records to electronic for pharmaceutical companies while being compliant
Following the rapid advances being made in the field of information management in the past quarter of a century or so, computers have increasingly come to replace paper as the source in which important documents are created and stored. Till the last decade of the previous century, organizations used paper to record and document information relating to their research, development and business. From around the start of the 1990’s; the shift towards computerization of paper…
Added by Adam Fleming on May 12, 2017 at 6:06am — No Comments
 Thorough understanding of the CERT program is necessary to fix error rates
Thorough understanding of the CERT program is necessary to fix error rates
The Comprehensive Error Rate Testing (CERT) is a program used by the Centers for Medicare & Medicaid Services (CMS) for calculating the rate or extent of improper payments of Medicare Fee-for-Service (FFS). The method used by the CMS under CERT is to pick up a stratified random sample of some 40,000 claims that are statistically valid and calculate and audit it to determine if the payments were made in accordance with the rules relevant to items such as coding, Medicare coverage, and…
ContinueAdded by Roger Steven on May 10, 2017 at 8:45am — No Comments
 What to expect during GMP Inspection
What to expect during GMP Inspection
Regulatory agencies such as the FDA conduct inspections for product-related Good Manufacturing Practice (GMP) in the course of assessing an application for marketing approval. The basic purpose of product-related GMP inspections is to check for compliance with the GMP guidelines set out in the International Conference on Harmonization (ICH) and national and local GMP regulations. GMP is the slightest standard that a manufacturer has to mandatorily meet in its production…
Added by Adam Fleming on April 3, 2017 at 6:29am — No Comments
 Right now Medical device hazard analysis, the core of medical devices
Right now Medical device hazard analysis, the core of medical devices
Medical device hazard analysis is of vital importance to a medical device. Medical device hazard analysis is at the heart of medical devices because if the device is not analyzed thoroughly for the hazard, or danger, that it poses, it is likely to cause problems of any kind to the user. Many a time, it becomes a matter of life and death. This is why medical device hazard analysis is of foremost importance.…
Added by Adam Fleming on March 27, 2017 at 8:31am — No Comments
 Supplier Management Conference for Medical Device Manufacturing in HONG KONG
Supplier Management Conference for Medical Device Manufacturing in HONG KONG
Overview:
Supplier selection and management is one of the critical issues for medical device manufacturers. Suppliers provide materials and services to the device manufacturer, which means that they can be critical to performance and delivery of your device. Neither the FDA nor your notified body regulates your suppliers (with…
ContinueAdded by John Robinson on February 1, 2017 at 4:55am — No Comments
 Water System Validation in Pharmaceuticals Industry 2017
Water System Validation in Pharmaceuticals Industry 2017
Overview:
This course is designed to provide a microbiology-focused education about all aspects of water systems and how biofilm manages to thrive there. Prior microbiological education or training, though a plus, is not a requirement because engineers and other non-biologists also need this training if they are involved with any aspect of water…
ContinueAdded by John Robinson on January 31, 2017 at 5:09am — No Comments
 Statistics for the Non-Statistician: Is it necessary?
Statistics for the Non-Statistician: Is it necessary?
Statistics for the non-statistician: Isn't this some kind of a contradiction in terms? Why does a non-statistician need to rake his head over statistics? Is statistics a life skill or a lifesaving one? If it is not, why is statistics for the non-statistician necessary? At first glance, statistics for the non-statistician may appear unnecessary and redundant,…
ContinueAdded by John Robinson on January 25, 2017 at 4:59am — No Comments
 Management for Medical Device Industry
Management for Medical Device Industry
A look at these gigantic figures perhaps gives some perspective of the importance of document management for the medical device industry: The US total market for medical devices is valued at over $110 billion annually. It makes up nearly two fifths of the global market, and is expected to grow by over 20 percent by 2016.
While many…
Added by John Robinson on January 23, 2017 at 7:46am — No Comments
Latest Blog Posts
- Know more about Wearable Medical Devices | Operon Strategist
- ISO 13485 - QMS Medical Device Certification Consultant
- Supplier Auditing for Medical Device Companies
- Why and How Do We Fail an FDA Inspection?
- The New EU Medical Device Regulation
- What do Your Customers Really Think About Your Complaint Handling?
- Will smart devices be the smartphones of the medical device industry?
Most Popular Blog Posts
- Medical Affairs Role in Pharmaceutical Companies
- Medical Affairs Services: Role in Planning of New Pharmaceutical Product Launch
- 10 Innovations That Will Transform Medicine ... Harvard Business Review
- IT’S A NO BRAINER! Action needed to stop children being exposed to chemicals that harm their brain development!
- ISO 13485 - QMS Medical Device Certification Consultant
- What do Your Customers Really Think About Your Complaint Handling?
- A Process Approach to Quality Management Systems
Monthly Archives
2024
- May (1)
2022
2020
2019
2018
- December (5)
- November (8)
- October (8)
- September (7)
- August (8)
- July (9)
- June (3)
- April (7)
- March (7)
- February (21)
- January (32)
2017
- December (11)
- November (39)
- October (64)
- September (60)
- August (34)
- July (30)
- June (22)
- May (40)
- April (16)
- March (12)
- February (18)
- January (15)
2016
2015
2014
2013
2012
- October (3)
- September (1)
- August (9)
- July (8)
- June (8)
- May (11)
- April (6)
- March (11)
- February (1)
- January (6)
2011
- December (2)
- November (3)
- October (7)
- September (3)
- July (4)
- June (4)
- April (7)
- March (4)
- February (3)
- January (7)
2010
- December (8)
- November (11)
- October (6)
- September (11)
- August (18)
- July (28)
- June (24)
- May (10)
- April (31)
- March (44)
- February (45)
- January (85)
2009
- December (104)
- November (63)
- October (44)
- September (68)
- August (53)
- July (37)
- June (50)
- May (43)
- April (63)
- March (106)
- February (36)
- January (15)
2008
1999
- November (5)
© 2024 Created by CC-Conrad Clyburn-MedForeSight.
Powered by
![]()

